
Haemoglobin
 المؤلف:
Peter Atkins, Tina Overton, Jonathan Rourke, Mark Weller, and Fraser Armstrong
المؤلف:
Peter Atkins, Tina Overton, Jonathan Rourke, Mark Weller, and Fraser Armstrong
 المصدر:
Shriver and Atkins Inorganic Chemistry ,5th E
المصدر:
Shriver and Atkins Inorganic Chemistry ,5th E
 الجزء والصفحة:
739
الجزء والصفحة:
739
 2025-10-23
2025-10-23
 52
52
Haemoglobin
Key point: Haemoglobin consists of a tetramer of myoglobin-like subunits, with four Fe sites that bind O2 cooperatively. Haemoglobin (Hb, 68 kg mol–1, Fig. 27.17) is the O2 transport protein found in special cells known as erythrocytes (red blood cells): a liter of human blood contains about 150 g of Hb. Simplistically, Hb can be thought of as a tetramer of myoglobin-like units with a cavity in the middle. There are in fact two types of Mb-like subunits, which differ slightly in their structures, and Hb is referred to as an a2 b2 tetramer. The O2 binding curves for Mb and Hb are shown in Fig. 27.18: it is highly significant that the curve for Hb is sigmoidal, which indicates that uptake and release of successive O2 molecules is cooperative. At low O2 partial pressure and greater acidity (as in venous blood and muscle tissue following aggressive exercise) Hb has low affinity for O2. This low affinity enables Hb to transfer its O2 to Mb. As the pressure increases, so does the affinity of Hb for O2 and as a result Hb can pick up O2 in the lungs. This change in affinity can be attributed to there being two conformations. The tensed state (T) has a low affinity and the relaxed state (R) has a high affinity. Deoxy-Hb is T and fully loaded oxy-Hb is R. To understand the molecular basis of cooperativity we need to refer to Fig. 27.15. Binding of the first O2 molecule to the T-state molecule is weak, but the decrease in the size of the Fe allows it to move into the plane of the porphyrin ring. This motion is particularly important for Hb because it pulls on the proximal histidine ligand and helix F moves. This movement is transmitted to the other O2 binding sites, the effect being to move the other Fe atoms closer to their respective ring planes and thereby convert the protein into the R state. The way is thereby opened for them to bind O2, which they now do with greater ease, although the statistical probability decreases as saturation is approached.
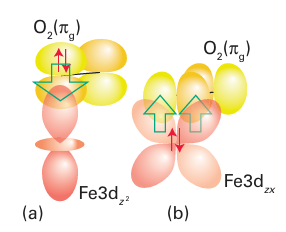
Figure 27.16 The orbitals used to form the Fe O2 adduct of myoglobin and haemoglobin. This model considers the O2 ligand to be in a singlet state, in which the full 2πg orbital donates an electron pair and the other 2πg orbital acts as a p-electron pair acceptor.
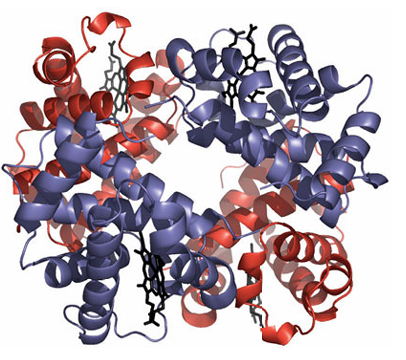
Figure 27.17 Haemoglobin is an α2β2 tetramer. Its and subunits are very similar to myoglobin.
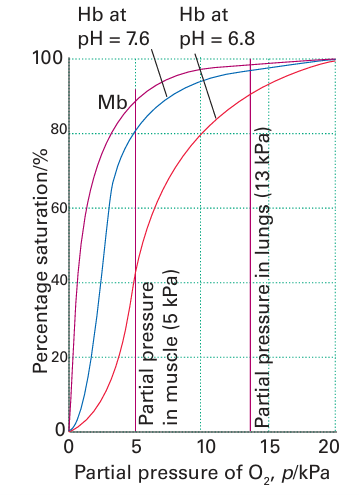
Figure 27.18 Oxygen binding curves for myoglobin and haemoglobin showing how cooperativity between the four sites in haemoglobin gives rise to a sigmoidal curve. The binding of the first O2 molecule to haemoglobin is unfavourable, but it results in a greatly enhanced affinity for subsequent O2 molecules.
 الاكثر قراءة في مواضيع عامة في الكيمياء العضوية
الاكثر قراءة في مواضيع عامة في الكيمياء العضوية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة


