


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 26-1-2017
Date: 28-12-2016
Date: 19-2-2019
|
Mass Relations from Equations
The relative numbers of reactant and product molecules (or the relative numbers of moles) are indicated by the coefficients of a balanced chemical equation. Using molar masses, we can compute the relative masses of reactants and products in a chemical reaction.
Example 1
Ammonia reacts with oxygen according to the equation

Thus 4 moles of NH3 react with 3 moles of O2 to form 2 moles of N2 and 6 moles of H2O. Suppose that the reaction consumes 10.00 g NH3. This corresponds
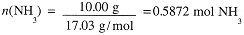
From the mole ratios, the reaction must involve
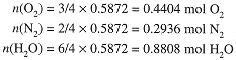
Using the molar masses, we can compute the mass of O2 reacting and the masses of N2 and H2O produced:
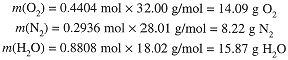
Notice that mass is conserved (as required) in the reaction:
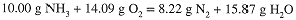
Often a problem gives the mass of one reactant or product and you are to determine the masses of the other substances involved. In problems of this kind, you may assume that sufficient amounts of the other reactants are present. If the masses of two reactants are given, you must determine which one (if either) is in excess. The best approach is to convert the given masses to moles and compare the mole ratio to that determined by the balanced equation.
Example 2
A solution containing 2.00 g of Hg(NO3)2 was added to a solution containing 2.00 g of Na2S. Calculate the mass of products formed according to the reaction

We begin by calculating the number of moles of each reactant:
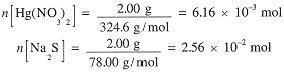
The equation indicates that equimolar quantities of reactants are required. Hence the given amount of Hg2(NO3)2 requires only 6.16 × 10-3 mol of Na2S. Thus 1.94 × 10-2 mol (1.51 g) of Na2S remains unreacted. Hg2(NO3)2 is the limiting reagent and we expect to form 6.16 × 10-3 mol HgS and 2(6.16 × 10-3) = 1.23 × 10-2 mol NaNO3:
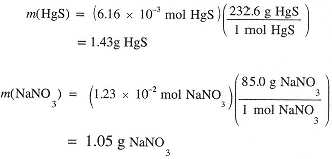
In summary, the original 4.00 g of reagents has been transformed to 1.43 g HgS and 1.05 g NaNO3 with 1.51 g Na2S unreacted. Note that the sum of the final masses is 3.99 g, within round-off error of the original 4.00 g.


|
|
|
|
دراسة: إجراء واحد لتقليل المخاطر الجينية للوفاة المبكرة
|
|
|
|
|
|
|
"الملح والماء" يمهدان الطريق لأجهزة كمبيوتر تحاكي الدماغ البشري
|
|
|
|
|
|
بالصور: عند زيارته لمعهد نور الإمام الحسين (عليه السلام) للمكفوفين وضعاف البصر في كربلاء.. ممثل المرجعية العليا يقف على الخدمات المقدمة للطلبة والطالبات
|
|
|
|
ممثل المرجعية العليا يؤكد استعداد العتبة الحسينية لتبني إكمال الدراسة الجامعية لشريحة المكفوفين في العراق
|
|
|
|
ممثل المرجعية العليا يؤكد على ضرورة مواكبة التطورات العالمية واستقطاب الكفاءات العراقية لتقديم أفضل الخدمات للمواطنين
|
|
|
|
العتبة الحسينية تستملك قطعة أرض في العاصمة بغداد لإنشاء مستشفى لعلاج الأورام السرطانية ومركز تخصصي للتوحد
|