علم الكيمياء

تاريخ الكيمياء والعلماء المشاهير

التحاضير والتجارب الكيميائية

المخاطر والوقاية في الكيمياء

اخرى

مقالات متنوعة في علم الكيمياء

كيمياء عامة


الكيمياء التحليلية

مواضيع عامة في الكيمياء التحليلية

التحليل النوعي والكمي

التحليل الآلي (الطيفي)

طرق الفصل والتنقية


الكيمياء الحياتية

مواضيع عامة في الكيمياء الحياتية

الكاربوهيدرات

الاحماض الامينية والبروتينات

الانزيمات

الدهون

الاحماض النووية

الفيتامينات والمرافقات الانزيمية

الهرمونات


الكيمياء العضوية

مواضيع عامة في الكيمياء العضوية

الهايدروكاربونات

المركبات الوسطية وميكانيكيات التفاعلات العضوية

التشخيص العضوي

تجارب وتفاعلات في الكيمياء العضوية


الكيمياء الفيزيائية

مواضيع عامة في الكيمياء الفيزيائية

الكيمياء الحرارية

حركية التفاعلات الكيميائية

الكيمياء الكهربائية


الكيمياء اللاعضوية

مواضيع عامة في الكيمياء اللاعضوية

الجدول الدوري وخواص العناصر

نظريات التآصر الكيميائي

كيمياء العناصر الانتقالية ومركباتها المعقدة


مواضيع اخرى في الكيمياء

كيمياء النانو

الكيمياء السريرية

الكيمياء الطبية والدوائية

كيمياء الاغذية والنواتج الطبيعية

الكيمياء الجنائية


الكيمياء الصناعية

البترو كيمياويات

الكيمياء الخضراء

كيمياء البيئة

كيمياء البوليمرات

مواضيع عامة في الكيمياء الصناعية

الكيمياء الاشعاعية والنووية
Compounds of boron with nitrogen
المؤلف:
Peter Atkins, Tina Overton, Jonathan Rourke, Mark Weller, and Fraser Armstrong
المصدر:
Shriver and Atkins Inorganic Chemistry ,5th E
الجزء والصفحة:
ص335-337
2025-08-31
50
Compounds of boron with nitrogen
Key points: Compounds containing BN, which is isoelectronic with CC, include the ethane analogue ammonia borane H3 NBH3, the benzene ana Logue H3N3B3H3, and BN analogues of graphite and diamond.
The thermodynamically stable phase of boron nitride, BN, consists of planar sheets of atoms like those in graphite (Section 14.5). The planar sheets of alternating B and N atoms consist of edge shared hexagons and, as in graphite, the B N distance within the sheet (145 pm) is much shorter than the distance between the sheets (333 pm, Fig. 13.4). The difference between the structures of graphite and boron nitride, however, lies in the register of the atoms of neighbouring sheets: in BN, the hexagonal rings are stacked directly over each other, with B and N atoms alternating in successive layers; in graphite, the hexagons are staggered. Molecular orbital calculations suggest that the stack ing in BN stems from a partial positive charge on B and a partial negative charge on N. This charge distribution is consistent with the electronegativity difference of the two elements (XP(B) = 2.04, XP(N) = 3.04).
As with impure graphite, layered boron nitride is a slippery material that is used as a lubricant. Unlike graphite, however, it is a colourless electrical insulator, as there is a large energy gap between the filled and vacant π bands. The size of the band gap is consistent with its high electrical resistivity and lack of absorption in the visible spectrum. In keeping with this large band gap, BN forms a much smaller number of intercalation compounds than graphite (Section 14.5). In contrast to graph-ite, layered boron nitride is stable in air up to 1000°C, making it a useful refractory material.
Layered boron nitride changes into a denser cubic phase at high pressures and temperatures (60 kbar and 2000C, Fig. 13.5).
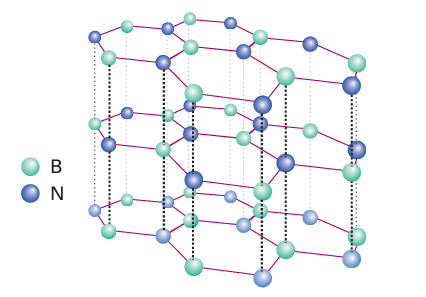
Figure 13.4 The structure of layered hexagonal boron nitride. Note that the rings are in register between layers.
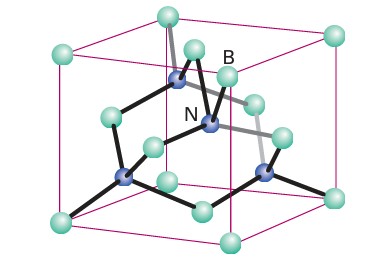
Figure 13.5 The sphalerite structure of cubic boron nitride.
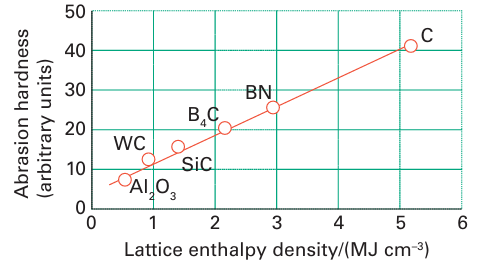
Figure 13.6 The correlation of hardness with lattice enthalpy density (the lattice enthalpy divided by the molar volume of the substance). The point for carbon represents diamond; that for boron nitride represents the diamond-like sphalerite structure.
This phase is a hard crystalline analogue of diamond but, as it has a lower lattice enthalpy, it has a slightly lower mechanical hardness (Fig. 13.6). Cubic boron nitride is manufactured and used as an abrasive for certain high-temperature applications in which diamond cannot be used because it forms carbides with the material being ground. The fact that BN and CC are isolectronic suggests that there might be analogies between these compounds and hydrocar bons. Many amine-boranes, the boron nitrogen analogues of saturated hydrocarbons, can be synthesized by reaction between a nitrogen Lewis base and a boron Lewis acid:
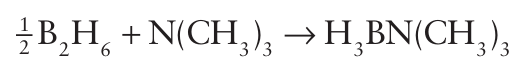
However, although amine-boranes are isoelectronic with hy drocarbons, their properties are significantly different, in large part due to the difference in electronegativities of B and N. For example, whereas ammoniaborane, H3 NBH3 , is a solid at room temperature with a vapour pressure of a few pascals, its ana logue ethane, H3CCH3, is a gas that condenses at 89C. This difference can be traced to the difference in polarity of the two molecules: ethane is nonpolar, whereas ammoniaborane has a large dipole moment of 5.2 D (20).
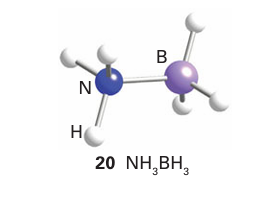
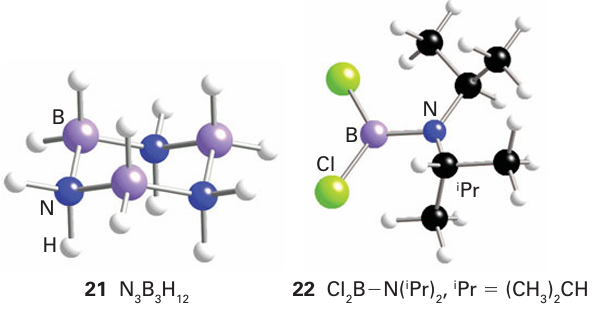
Several BN analogues of the amino acids have been prepared, including ammoniacarboxyborane, H3 NBH2 COOH, the ana logue of propionic acid, CH3 CH2 COOH. These compounds display significant physiological activity, including autoinhibition and reduction of serum cholesterol. The simplest unsaturated boron nitrogen compound is amino borane, H2 NBH2 , which is isoelectronic with ethene. It has only a transient existence in the gas phase because it readily forms cyclic ring compounds such as a cyclohexane analogue (21). However, the amino boranes do survive as monomers when the double bond is shielded from reaction by bulky alkyl groups on the N atom and by Cl atoms on the B atom (22). For instance, monomeric amino boranes can be synthesized readily by the reaction of a dialkylamine and a boron halide:
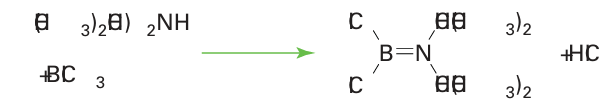
The reaction also occurs with xylyl (2,4,6-trimethylphenyl) groups in place of isopropyl groups.
Apart from layered boron nitride, the best-known unsaturated compound of boron and nitrogen is borazine, B3 N3 H6 (23), which is isoelectronic and isostructural with benzene. Borazine was first prepared by Alfred Stock in 1926 by the reaction between diborane and ammonia. Since then, many symmetrically trisubstituted derivatives have been made by procedures that depend on the protolysis of BCl bonds of BCl3 by an ammonium salt (24):
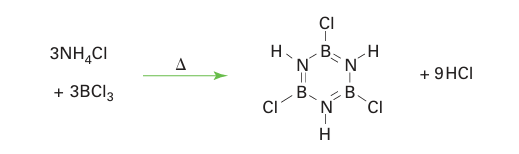
The use of an alkylammonium chloride yields N-alkyl substi-tutedB-trichloroborazines.
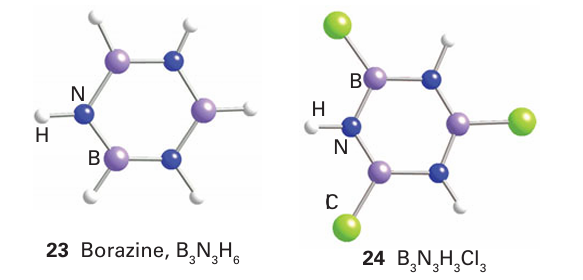
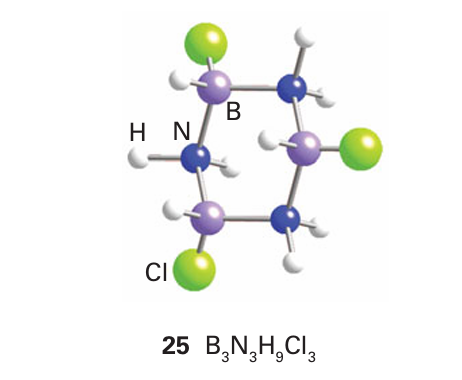
Despite their structural resemblance, there is little chemical resemblance between borazine and benzene. Once again, the difference in the electronegativities of boron and nitrogen is influential, and BCl bonds in trichloroborazine are much more labile than the CCl bonds in chlorobenzene. In the borazine compound, the π electrons are concentrated on the N atoms and there is a partial positive charge on the B atoms that leaves them open to electrophilic attack. A sign of the difference is that the reaction of a chloroborazine with a Grignard reagent or hydride source results in the substitution of Cl by alkyl, aryl, or hydride groups. Another example of the difference is the ready addition of HCl to borazine to produce a trichlorocyclohexane analogue (25):
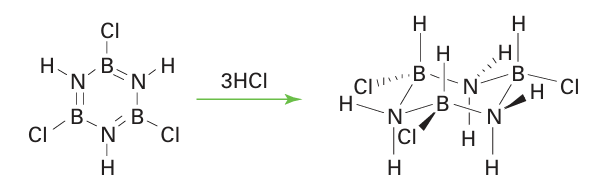
The electrophile, H, in this reaction attaches to the partially negative N atom and the nucleophile Cl attaches to the partially positive B atom.

 الاكثر قراءة في مواضيع عامة في الكيمياء العضوية
الاكثر قراءة في مواضيع عامة في الكيمياء العضوية
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام) قسم الشؤون الفكرية يصدر مجموعة قصصية بعنوان (قلوب بلا مأوى)
قسم الشؤون الفكرية يصدر مجموعة قصصية بعنوان (قلوب بلا مأوى)