


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
أقرأ أيضاً
التاريخ: 30-9-2020
التاريخ: 10-7-2018
التاريخ: 30-9-2020
التاريخ: 27-8-2018
|
The alkynes behave in many ways as if they were doubly unsaturated alkenes. For example, bromine adds to ethyne in two stages - first to give trans-1,2-dibromoethene by antarafacial addition, and finally to give 1,1,2,2-tetrabromoethane:
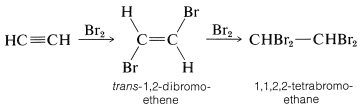
Likewise, anhydrous hydrogen fluoride adds first to give fluoroethene and ultimately to give 1,1-difluoroethane:
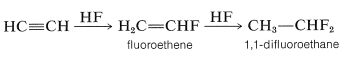
However, there is an interesting contrast in reactivity. Alkynes are substantially less reactive than corresponding alkenes toward many electrophiles. This is perhaps surprising because the electrons of a triple bond, like those of a double bond, are highly exposed, which suggests that the reactivity (nucleophilicity) of a triple bond should be high. Evidently this is not the case. A simple but reasonable explanation is that the carbocation formed from the alkyne is less stable than that from the alkene because it cannot achieve the sp2 hybrid-orbital configuration expected to be the most stable arrangement for a carbocation:
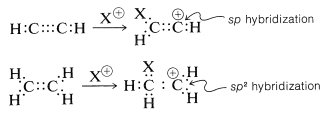


|
|
|
|
كيف تعزز نمو الشعر الصحي؟
|
|
|
|
|
|
|
10 فحوصات مهمة يجب القيام بها لسيارتك قبل الصيف
|
|
|
|
|
|
قسم التربية والتعليم ينظّم جلسةً حوارية لملاكه حول تأهيل المعلّمِين الجدد
|
|
|
|
جامعة العميد تحدّد أهداف إقامة حفل التخرّج لطلبتها
|
|
|
|
جامعة العميد تحتفي بتخرّج الدفعة الثانية من طلبة كلّية الطبّ
|
|
|
|
قسم الشؤون الفكريّة يشارك في المؤتمر العلمي الدولي الخامس في النجف الأشرف
|