


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 13-4-2019
Date: 30-1-2017
Date: 12-7-2017
|
Skipping liquids: Sublimation
Most substances go through the logical progression from solid to liquid to gas as they’re heated (or vice versa as they’re cooled). But a few substances go directly from the solid to the gaseous state without ever becoming a liquid. Scientists call this process sublimation. Dry ice — solid carbon dioxide, written as CO2(s) — is the classic example of sublimation. You can see dry ice pieces becoming smaller as the solid begins to turn into a gas, but no liquid forms during this phase change. The process of sublimation of dry ice is represented as
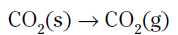
Besides dry ice, mothballs and certain solid air fresheners also go through the process of sublimation. The reverse of sublimation is deposition — going directly from a gaseous state to a solid state.


|
|
|
|
كيف تعزز نمو الشعر الصحي؟
|
|
|
|
|
|
|
10 فحوصات مهمة يجب القيام بها لسيارتك قبل الصيف
|
|
|
|
|
|
كلية الصيدلة في جامعة العميد تحتفي بتخرج الدفعة الأولى من طلبتها
|
|
|
|
قسم شؤون المعارف يعلن عن استمراره باستلام بحوث مؤتمر الإمام الكاظم (عليه السلام)
|
|
|
|
قسم الشؤون الفكرية يعلن عن تنظيم دورة تخصّصية حول التوحيد والشبهات المعاصرة
|
|
|
|
وفد حكومي من ميسان يطّلع على مشاتل العبّاسية
|